As stated in the World Health Organisation’s report of April 1, 2021, there were a total of 229 million cases of malaria and 409 000 deaths due to it, worldwide in 2019. WHO’s report also states that the African region contributes to 94% of the global burden. Children are the most vulnerable to infection and account for 67% of Malaria deaths worldwide. The approval for widespread use of the world’s first Malaria vaccine by WHO is thus a celebratory announcement for all.
On Wednesday, 7th October, the WHO approved the widespread use of RTS, S/AS01 malaria vaccine, the first in the world to fight against mosquito-borne disease. Although the vaccine does not guarantee complete immunity, prevention, or cure from the infection, it does promise to reduce the severity of infection.
The vaccine was developed some 6 years back and guaranteed to prevent 40% of malaria infections and 30% of severe cases. A more wide and rigorous pilot immunisation programme has been carried out by the researchers since 2019 in Ghana, Kenya, and Malawi in order to be able to apply for the approval of its widespread use.
The first dose of the Malaria vaccine has been administered to more than 800,000 children and no safety concerns have been reported, the WHO said. The organisation has now recommended that those children belonging to sub-Saharan Africa and in regions having moderate to high malaria transmission should be given four doses of the vaccine up to the age of two years.
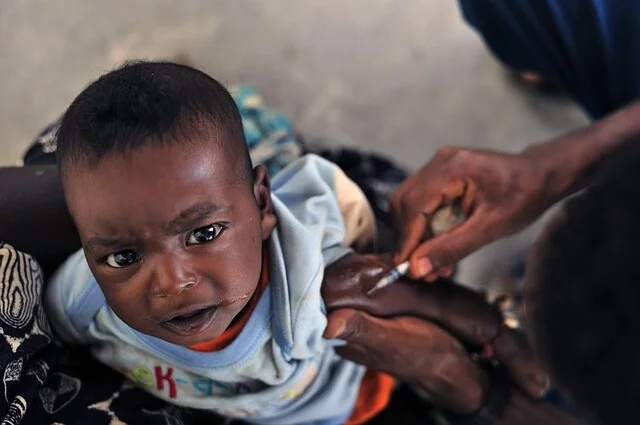
Although the efficacy of the Malaria vaccine is relatively low, it is yet a commendable success as it does have the potential to bring down the number of cases by 40% globally. This indicates that it will definitely save millions of lives that were lost previously due to a lack of proper medication.
The vaccine is yet to be made available to the world as its distribution cost and pricing need to be decided. The vaccine is developed by pharmaceutical giant GSK. The company has agreed to supply the vaccine at manufacturing cost plus 5% but hasn’t put forth a final figure.
Till now the company has donated a total of 10 million doses for the pilot study in Ghana, Kenya, and Malawi. The company has set a target for itself to provide 15 million doses each year. If all goes well and sufficient funds get arranged, the company might also start making the vaccines available for wider use from the end of 2022 or early 2023.
This count of the Malaria vaccine isn’t enough given the number of infections. The company might have to manufacture up to 100 million doses every year.








